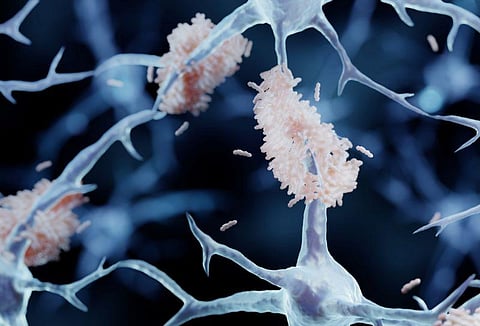
WEDNESDAY, May 3, 2023 (HealthDay News) -- Another experimental drug meant for Alzheimer disease looks so promising that drugmaker Eli Lilly plans to ask the U.S. Food and Drug Administration for full approval by the end of June.
Known as donanemab, the medication clears amyloid plaque from the brain. In a clinical trial, the drug slowed memory and thinking declines in early symptomatic Alzheimer disease patients by more than a third, Lilly said Wednesday. About 47 percent of those taking the medication had no decline on a key measure of thinking over a year compared with 29 percent of patients on a placebo.
But there were some risks noted in the results. The Lilly trial involved 1,700 patients, three of whom died during the study. Two of those deaths were attributed to brain swelling or microbleeds called amyloid-related imaging abnormalities.
About 52 percent of the patients in the trial were able to stop taking the medication by one year because of its effectiveness. About 72 percent could do so by a year and a half, the company said. In the trial, a group with intermediate levels of tau had a 35 percent slowing in cognitive and functional decline. In the intermediate group combined with a group with higher tau levels, slowing in decline was 22 percent.
"We are extremely pleased that donanemab yielded positive clinical results with compelling statistical significance for people with Alzheimer's disease in this trial," Daniel Skovronsky, M.D., Lilly chief scientific and medical officer and president of Lilly Research Laboratories, said in a company news release. "This is the first Phase 3 trial of any investigational medicine for Alzheimer's disease to deliver 35 percent slowing of clinical and functional decline."
More Information